Chemistry, 12.05.2021 04:30 irabrooks14
To lower the chance of suffering from decompression sickness (the bends), scuba divers use a mixture of gases in their air tank (typically oxygen and nitrogen gas in recreational dives). Assuming no other gas is present besides oxygen and nitrogen, if the mole fraction of oxygen present is 0.21, what is the partial pressure of nitrogen gas if the total pressure is 111.7 atm

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 19:00
Avolleyball player hit a ball with a mass of 0.25 kg. the average acceleration of the ball is 15.5 m/s². how much force did the volleyball player apply to the ball? 62.0 n 3.87 n 62.0 m/s² 3.87 m/s²
Answers: 2

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3
You know the right answer?
To lower the chance of suffering from decompression sickness (the bends), scuba divers use a mixture...
Questions


World Languages, 25.01.2022 23:30

Mathematics, 25.01.2022 23:30



Computers and Technology, 25.01.2022 23:40

Mathematics, 25.01.2022 23:40


Chemistry, 25.01.2022 23:40

World Languages, 25.01.2022 23:40

Mathematics, 25.01.2022 23:40


Social Studies, 25.01.2022 23:40







Mathematics, 25.01.2022 23:50

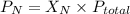
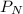 = partial pressure of nitrogen
= partial pressure of nitrogen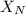 = mole fraction of nitrogen
= mole fraction of nitrogen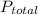 = total pressure
= total pressure