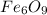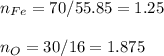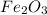Chemistry, 13.05.2021 05:30 sarinaneedshelp01
A compound is 70.0% iron and 30.0% oxygen. Its molecular mass is 479.1 g/mol. Determine the molecular formula.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Which of the following can be used to measure electricity
Answers: 1

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 23.06.2019 00:30
What would be the original temperature of a gas that has a volume of 2.0 l and a pressure of 2.0 atm and an unknown temperature that the volume increased to 3.5 l in its pressure decreased to 1.0 atm if the final temperature is measured to be 11°c
Answers: 1
You know the right answer?
A compound is 70.0% iron and 30.0% oxygen. Its molecular mass is 479.1 g/mol. Determine the
molecul...
Questions


Mathematics, 13.07.2019 15:00

English, 13.07.2019 15:00



Mathematics, 13.07.2019 15:00


History, 13.07.2019 15:00

Mathematics, 13.07.2019 15:00

History, 13.07.2019 15:00

Mathematics, 13.07.2019 15:00

Computers and Technology, 13.07.2019 15:00




Mathematics, 13.07.2019 15:00


Arts, 13.07.2019 15:00