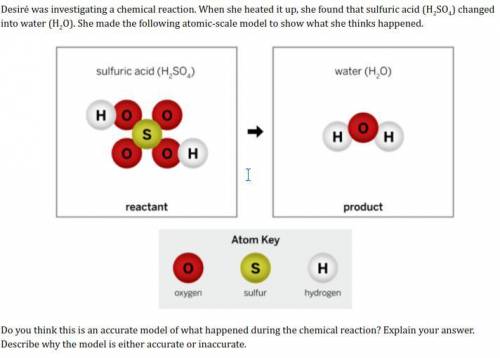
HELP please need this done asap
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 16:00
How do dying stars contribute to the formation of planets
Answers: 1

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

You know the right answer?
Questions



Mathematics, 14.01.2021 23:10

Biology, 14.01.2021 23:10



Mathematics, 14.01.2021 23:10

History, 14.01.2021 23:10

Mathematics, 14.01.2021 23:10

Physics, 14.01.2021 23:10

Health, 14.01.2021 23:10





Geography, 14.01.2021 23:10

Mathematics, 14.01.2021 23:10


Mathematics, 14.01.2021 23:10

Chemistry, 14.01.2021 23:10