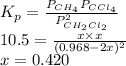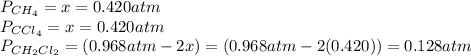Chemistry, 14.05.2021 03:00 karose4590
The equilibrium constant, Kp, for the following reaction is 10.5 at 350 K: 2CH2Cl2(g) CH4(g) CCl4(g) Calculate the equilibrium partial pressures of

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Aside from human impact, which of the following causes less water vapor production over a small area? (2 pderivartin
Answers: 1

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1
You know the right answer?
The equilibrium constant, Kp, for the following reaction is 10.5 at 350 K: 2CH2Cl2(g) CH4(g) CCl4(g)...
Questions

Computers and Technology, 19.10.2019 09:00


History, 19.10.2019 09:00



Mathematics, 19.10.2019 09:00

Mathematics, 19.10.2019 09:00

Mathematics, 19.10.2019 09:00







History, 19.10.2019 09:00


History, 19.10.2019 09:00

Mathematics, 19.10.2019 09:00


Mathematics, 19.10.2019 09:00

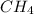 ,
, 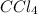 and
and 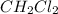 is 0.420 atm, 0.420 atm and 0.128 atm.
is 0.420 atm, 0.420 atm and 0.128 atm.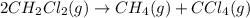
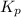 for this reaction is as follows.
for this reaction is as follows.