
The heat of combustion per mole for acetylene, C2H2(g), is -1299.5 kJ/mol. Assuming that the combustion products are CO2(g) and H2O(l), and given that the enthalpy of formation is -393.5 kJ/mol for CO2(g) and -285.8 kJ/mol for H2O(l), find the enthalpy of formation of C2H2(g).

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1

You know the right answer?
The heat of combustion per mole for acetylene, C2H2(g), is -1299.5 kJ/mol. Assuming that the combust...
Questions

Mathematics, 23.12.2019 03:31







Biology, 23.12.2019 03:31

Mathematics, 23.12.2019 03:31


English, 23.12.2019 03:31


History, 23.12.2019 03:31

Mathematics, 23.12.2019 03:31

Biology, 23.12.2019 03:31

Spanish, 23.12.2019 03:31

Advanced Placement (AP), 23.12.2019 03:31

Mathematics, 23.12.2019 03:31

Mathematics, 23.12.2019 03:31

History, 23.12.2019 03:31

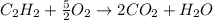
![\Delta H^{o}_{rxn} = \sum \Delta H_{products} - \sum \Delta H_{reactants}\\\Delta H^{o}_{rxn} = [2\Delta H^{o}_{f}(CO_{2}) + \Delta H^{o}_{f} (H_{2}O)] - [\Delta H^{o}_{f}(C_{2}H_{2}) + \frac{5}{2} \Delta H^{o}_{f} O_{2}]\\-1299.5 = 2(-393.5) + (-285.8) - \Delta H^{o}_{f} (C_{2}H_{2})\\\Delta H^{o}_{f} (C_{2}H_{2}) = 227.7 kJ/mol](/tpl/images/1323/2810/92eb3.png)


