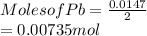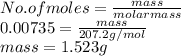When a lead acid car battery is recharged by the alternator, it acts essentially as an electrolytic cell in which solid lead(II) sulfate PbSO4 is reduced to lead at the cathode and oxidized to solid lead(II) oxide PbO at the anode. Suppose a current of 62.0 is fed into a car battery for 23.0 seconds. Calculate the mass of lead deposited on the cathode of the battery. Round your answer to 3 significant digits. Also, be sure your answer contains a unit symbol.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 07:30
All cells are made of four types of acids, lipids, proteins, and carbohydrates.
Answers: 1

Chemistry, 23.06.2019 07:00
What are the trends and exceptions to the trends in electron affinity?
Answers: 1

Chemistry, 23.06.2019 10:00
What is the density, d, of a substance with a volume of v = 12.5 cm3 and a mass of m = 74.4 g ?
Answers: 1
You know the right answer?
When a lead acid car battery is recharged by the alternator, it acts essentially as an electrolytic...
Questions



Mathematics, 21.11.2019 08:31


Biology, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31

Social Studies, 21.11.2019 08:31


Mathematics, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31

Biology, 21.11.2019 08:31

Computers and Technology, 21.11.2019 08:31


English, 21.11.2019 08:31




Social Studies, 21.11.2019 08:31


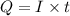
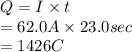
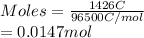
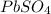 is 2. So, moles deposited by Pb is as follows.
is 2. So, moles deposited by Pb is as follows.