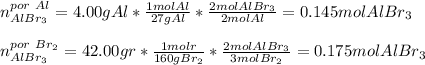Chemistry, 15.05.2021 06:10 dropnsocks8315
Se hace reaccionar 4,00 g de aluminio y 42,00 g de bromo, según la reacción: Al(s)+Br2(l)⟶AlBr3(s) Calcular las moles de AlBr3(s) producido. PA: Al=27; Br=80

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2
You know the right answer?
Se hace reaccionar 4,00 g de aluminio y 42,00 g de bromo, según la reacción: Al(s)+Br2(l)⟶AlBr3(s) C...
Questions



Mathematics, 06.11.2020 06:00

English, 06.11.2020 06:00

Chemistry, 06.11.2020 06:00



Mathematics, 06.11.2020 06:00

Mathematics, 06.11.2020 06:00


Mathematics, 06.11.2020 06:00


Chemistry, 06.11.2020 06:00



Chemistry, 06.11.2020 06:00



History, 06.11.2020 06:00

Mathematics, 06.11.2020 06:00