Chemistry, 17.05.2021 04:00 katrinajernae8216
A student performed the following reaction in his Chemistry class: Zn + 2HCl ---> ZnCl2+H2. He started with 32.70 g of Zn metal and 18.23 g HCl and got 60.1 g of of ZnCl2.
(I) How much ZnC12 should he have produced?
(II) What was his percentage yield?
(III) If the 32.70 g of Zinc that he started with contained impurities (materials other than Zinc), will this support the student's percentage yield?
Explain briefly.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Your friend offers to show you an intrusive igneous rock. which of the following would you expect to see?
Answers: 1

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

Chemistry, 22.06.2019 15:00
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1
You know the right answer?
A student performed the following reaction in his Chemistry class: Zn + 2HCl ---> ZnCl2+H2. He st...
Questions

Mathematics, 01.02.2021 20:00


Mathematics, 01.02.2021 20:00



Mathematics, 01.02.2021 20:00

Mathematics, 01.02.2021 20:00


Mathematics, 01.02.2021 20:00

Social Studies, 01.02.2021 20:00

Mathematics, 01.02.2021 20:00


Chemistry, 01.02.2021 20:00


English, 01.02.2021 20:00

Computers and Technology, 01.02.2021 20:00

Mathematics, 01.02.2021 20:00


Social Studies, 01.02.2021 20:00

Mathematics, 01.02.2021 20:00

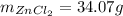
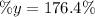
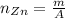
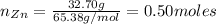
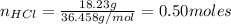
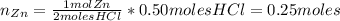
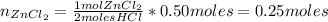

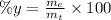
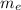 : is the experimental mass = 60.1 g
: is the experimental mass = 60.1 g 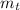 : is the theoretical mass = 34.07 g
: is the theoretical mass = 34.07 g