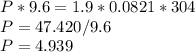Chemistry, 17.05.2021 17:40 loveyeti106838
At 304 K, a 9.6 L tank contains 1.9 moles of H2 gas under an unknown pressure in atm. What is the pressure of the gas in the tank? (R = 0.0821 L*atm/mol*K)

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2

Chemistry, 23.06.2019 03:00
Select the correct answer. wax is a nonpolar substance. in which type of substance is it most soluble?
Answers: 2
You know the right answer?
At 304 K, a 9.6 L tank contains 1.9 moles of H2 gas under an unknown pressure in atm. What is the pr...
Questions



History, 01.12.2021 03:20



Engineering, 01.12.2021 03:20





Health, 01.12.2021 03:20


Biology, 01.12.2021 03:20

History, 01.12.2021 03:20

Physics, 01.12.2021 03:20



Computers and Technology, 01.12.2021 03:20