
Chemistry, 18.05.2021 17:50 Tonyang1742
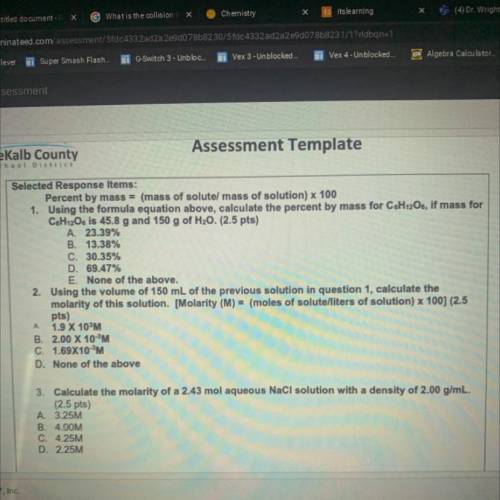
Percent by mass = (mass of solutel mass of solution) x 100
1. Using the formula equation above, calculate the percent by mass for C&H12O6, if mass for
C6H12Os is 45.8 g and 150 g of H20. (2.5 pts)
A. 23.39%
B. 13.38%
C. 30.35%
D. 69.47%
E. None of the above.
2. Using the volume of 150 mL of the previous solution in question 1, calculate the
molarity of this solution. [Molarity (M) = (moles of solute/liters of solution) x 100] (2.5
pts)
1.9 X 10%M
B. 2.00 X 10 M
C. 1.69X10M
D. None of the above
A
3. Calculate the molarity of a 2.43 mol aqueous NaCl solution with a density of 2.00 g/mL.
(2.5 pts)
A 3.25M
B. 4.00M
C. 4.25M
D. 2.25M

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 08:30
Agroup of students is studying convection current. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other is in an area with warm air. after 10 minutes, the balloon are released from a height of 1 meter. which of the following to the students most likely observe? a) the warm balloon expands and rises. the cold balloon shrinks and sinks b) the balloon both rise. the cold balloon is larger than the warm balloon c) the cold balloon expands and rises. the warm balloon shrinks and sinks d) the balloon rise at the same rate. both balloons are the same size
Answers: 1


Chemistry, 22.06.2019 10:40
If an area has high air pressure and low humidity, what type of weather will it most likely have? plz !
Answers: 1
You know the right answer?
Percent by mass = (mass of solutel mass of solution) x 100
1. Using the formula equation above, cal...
Questions







Biology, 19.11.2020 18:40

Computers and Technology, 19.11.2020 18:40

Physics, 19.11.2020 18:40

Mathematics, 19.11.2020 18:40


Mathematics, 19.11.2020 18:40





English, 19.11.2020 18:40





