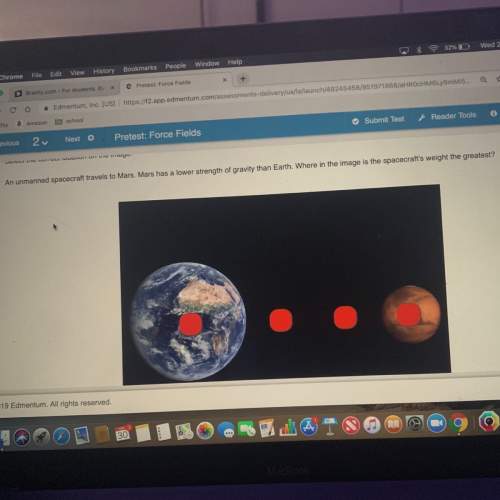
What is the concentration of HCl if 2.5L of 0.30 M NaOH
neutralizes 6.OL HCI?
NaOH...

Chemistry, 18.05.2021 21:00 moningersavannah
What is the concentration of HCl if 2.5L of 0.30 M NaOH
neutralizes 6.OL HCI?
NaOH + HCI --> H20 + NaCl

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 10:00
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 14:30
Ahypothesis must be testable and falsifiable to be considered scientific a. trueb. false
Answers: 1

You know the right answer?
Questions

History, 02.07.2019 14:40

History, 02.07.2019 14:40

History, 02.07.2019 14:40



Business, 02.07.2019 14:40

Social Studies, 02.07.2019 14:40

Mathematics, 02.07.2019 14:40


Business, 02.07.2019 14:40

Business, 02.07.2019 14:40



English, 02.07.2019 14:40


Business, 02.07.2019 14:40

Biology, 02.07.2019 14:40

History, 02.07.2019 14:40

History, 02.07.2019 14:40

Biology, 02.07.2019 14:40