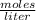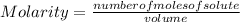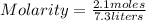Chemistry, 22.05.2021 06:20 jasjas3722
If 2.1 moles of NaCl is dissolved in a solution with a total volume of 7.3 liters, what is the molarity of the solution? Round to two decimals.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
If 1.63 times 10 negative 4 of helium dissolves in 100.0g of water, what is the concentration in parts per million
Answers: 3

Chemistry, 22.06.2019 02:10
Determine the percent sulfuric acid by mass of a 1.61 m aqueous solution of h2so4. %
Answers: 2

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2
You know the right answer?
If 2.1 moles of NaCl is dissolved in a solution with a total volume of 7.3 liters, what is the molar...
Questions

Mathematics, 28.06.2019 18:00



Geography, 28.06.2019 18:00



Mathematics, 28.06.2019 18:00


Mathematics, 28.06.2019 18:00




History, 28.06.2019 18:00


Biology, 28.06.2019 18:00