
Chemistry, 22.05.2021 19:50 kashusledbetter
Use the periodic table to determine what element cesium-135 (135/55C3) becomes after ... answers for most questions on finals chem test (10th)
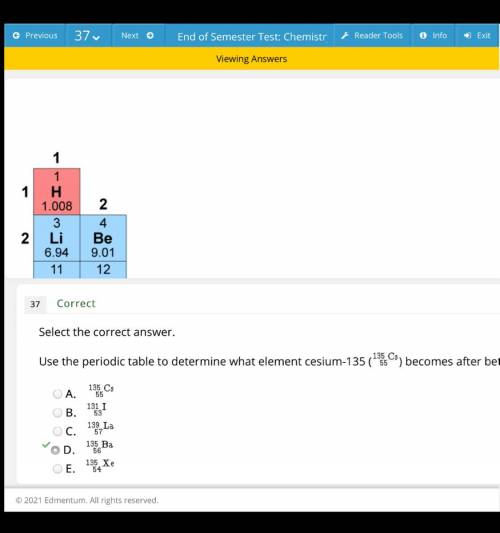

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
Use the periodic table to determine what element cesium-135 (135/55C3) becomes after ...
answers fo...
Questions

English, 13.07.2019 00:00



Health, 13.07.2019 00:00

English, 13.07.2019 00:00

Mathematics, 13.07.2019 00:00

History, 13.07.2019 00:00

English, 13.07.2019 00:00



History, 13.07.2019 00:00

Physics, 13.07.2019 00:00




History, 13.07.2019 00:00


History, 13.07.2019 00:00


Biology, 13.07.2019 00:00



