
PLEASE HELP!
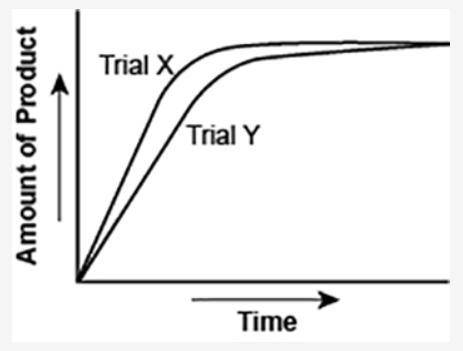
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different concentration of reactant was used during each trial, whereas the other factors were kept constant.
(I've attached the graph.)
Which of the following statements explains which trial has a lower concentration of the reactant?
a. Trial X, because the final volume of product formed is lower than Trial Y.
b. Trial X, because this reaction was initially fast and later stopped completely.
c. Trial Y, because the reaction was initially slow and later stopped completely.
d. Trial Y, because the volume of product formed per unit time is lower than Trial X.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
All cells are made of four types of acids, lipids, proteins, and carbohydrates.
Answers: 1

Chemistry, 22.06.2019 09:00
Look at the spectrums of a star moving towards earth and a motionless star. which of these is a correct inference that can be draw from the observation of the two spectrums? (2 points) the spectrum of a motionless star is difficult to be viewed separately using oridinary telescopes. the spectrum of a motionless star is identical to the spectrum of a star which moves towards earth. the spectrum of a star shifts towards the red region when the star moves towards earth. the spectrum of a star shifts towards the blue region when the star moves towards earth.
Answers: 2

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

You know the right answer?
PLEASE HELP!
The graph shows the volume of a gaseous product formed during two trials of a reaction...
Questions

Mathematics, 16.07.2019 02:30

Mathematics, 16.07.2019 02:30

Chemistry, 16.07.2019 02:30

Mathematics, 16.07.2019 02:30

Mathematics, 16.07.2019 02:30

History, 16.07.2019 02:30


Mathematics, 16.07.2019 02:30

Mathematics, 16.07.2019 02:30


Mathematics, 16.07.2019 02:30




History, 16.07.2019 02:30




Business, 16.07.2019 02:30



