Chemistry, 24.05.2021 09:10 alexkrol30083
A 5.00 mL sample of hydrochloric acid is titrated with 0.1293 M ammonia (a base). If the titration required 28.15 mL of ammonia, determine the following:
the original concentration of the acid
the original pH of the acid

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 12:30
Clyde and marilyn are riding a roller coaster. during which section(s) of the track is their potential energy converted to kinetic energy? a. from point b to point c only b. from point b to point d only c. from point a to point b only d. from point a to point b and from point c to point d
Answers: 1

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1
You know the right answer?
A 5.00 mL sample of hydrochloric acid is titrated with 0.1293 M ammonia (a base). If the titration r...
Questions




















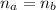

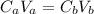
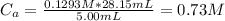
![pH = -log([H^{+}])](/tpl/images/1343/3808/0d4b9.png)