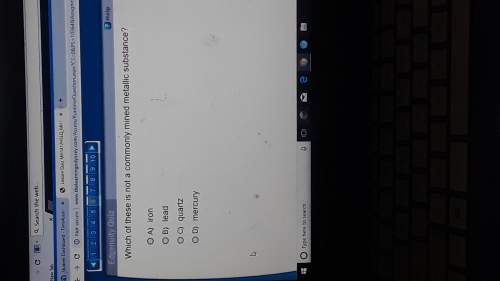
Chemistry, 26.05.2021 14:00 oliviajewelwilliams
1. Phosphorous reacts with bromine to form phosphorous tribromide. If 35.0 grams of bromine
reacted and 27.9 grams of phosphorous tribromide are actually formed.
what is the percent yield of phosphorous tribromide (PBr3)?
2 P + 3 Br2 ---> 2 PBr3
(Note that the molar masses: P=30.97g, Br = 79.90g) Show your steps
*
Please help me

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2


Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3
You know the right answer?
1. Phosphorous reacts with bromine to form phosphorous tribromide. If 35.0 grams of bromine
reacted...
Questions





Mathematics, 15.11.2019 23:31

Mathematics, 15.11.2019 23:31


English, 15.11.2019 23:31




Mathematics, 15.11.2019 23:31

Mathematics, 15.11.2019 23:31

Social Studies, 15.11.2019 23:31

History, 15.11.2019 23:31


Mathematics, 15.11.2019 23:31

Mathematics, 15.11.2019 23:31


Physics, 15.11.2019 23:31