
Chemistry, 27.05.2021 03:50 erenackermanlevijaeg
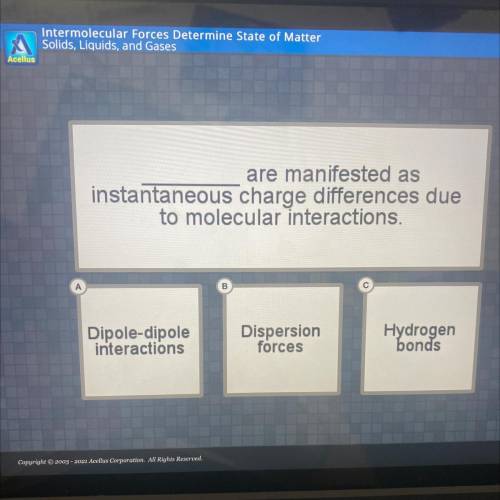
Are manifested as
instantaneous charge differences due
to molecular interactions.
Dipole-dipole
interactions
Dispersion
forces
Hydrogen
bonds

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
You know the right answer?
Are manifested as
instantaneous charge differences due
to molecular interactions.
Dipol...
to molecular interactions.
Dipol...
Questions



Mathematics, 04.12.2020 21:10





Computers and Technology, 04.12.2020 21:10




Mathematics, 04.12.2020 21:10

Mathematics, 04.12.2020 21:10

Mathematics, 04.12.2020 21:10

Mathematics, 04.12.2020 21:10

Mathematics, 04.12.2020 21:10


Mathematics, 04.12.2020 21:10




