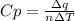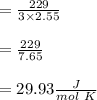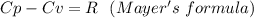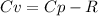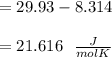Chemistry, 27.05.2021 05:10 genyjoannerubiera
When 229 J of energy is supplied as heat at constant pressure to 3.0mol Ar(g) the temperature of the sample increases by 2.55K. Calculate the molar heat capacities at constant volume and constant pressure of the gas.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 22.06.2019 12:00
Marcel just purchased 1.69 grams of iron fillings in order to make living putty for his 6 year old niece. how many moles of iron are made in his sample?
Answers: 1

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

You know the right answer?
When 229 J of energy is supplied as heat at constant pressure to 3.0mol Ar(g) the temperature of the...
Questions

Social Studies, 17.01.2020 18:31

Mathematics, 17.01.2020 18:31

Biology, 17.01.2020 18:31









Arts, 17.01.2020 18:31

English, 17.01.2020 18:31

Chemistry, 17.01.2020 18:31




History, 17.01.2020 18:31

History, 17.01.2020 18:31

Biology, 17.01.2020 18:31

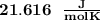 ".
".