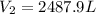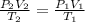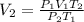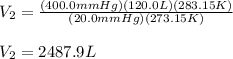Chemistry, 27.05.2021 06:30 qveenjordan6456
1. A volume of 120.0 liters of a gas is prepared at a pressure of 400.0 mm Hg and a temperature
100.0 °C. The gas is placed into a tank under high pressure. When the tank cools to 10.0°C,
the pressure of the gas is 20.0 mm Hg. What is the volume of the gas?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 14:30
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1


Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3
You know the right answer?
1. A volume of 120.0 liters of a gas is prepared at a pressure of 400.0 mm Hg and a temperature
100...
Questions







English, 13.02.2021 01:20


Mathematics, 13.02.2021 01:20

Mathematics, 13.02.2021 01:20

Mathematics, 13.02.2021 01:20

History, 13.02.2021 01:20

Mathematics, 13.02.2021 01:20