Chemistry, 27.05.2021 14:00 Shadow0202
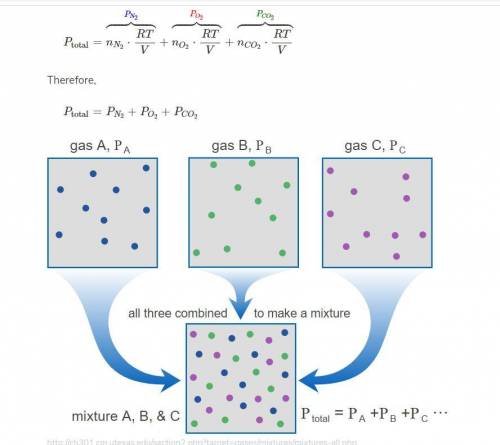
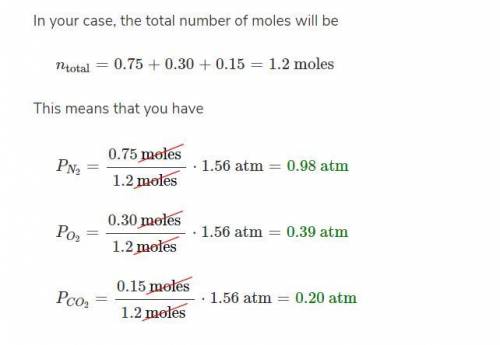
A mixture of oxygen( O2), dinitrogen monoxide (N2O), and argon (Ar) has a total pressure of 0.98 atm. What is the partial pressure of N2O, if the partial pressure of O2 is 0.48 atm and the partial pressure of Ar is 0.15 atm?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
At room temperature what happens to the average kinetic energy of the molecules of a solid, liquid, and a gas
Answers: 2



Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2
You know the right answer?
A mixture of oxygen( O2), dinitrogen monoxide (N2O), and argon (Ar) has a total pressure of 0.98 atm...
Questions

English, 04.10.2021 19:10

SAT, 04.10.2021 19:10

Biology, 04.10.2021 19:10

History, 04.10.2021 19:10



History, 04.10.2021 19:10

History, 04.10.2021 19:10




Physics, 04.10.2021 19:10








English, 04.10.2021 19:10