
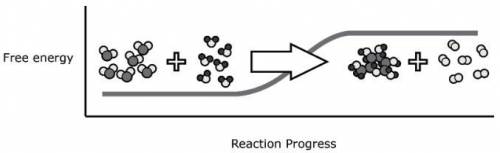
The diagram provided here shows the reactants and the products of a common chemical reaction. The line on the graph behind them represents the energy of a system as the reaction progresses. Which of the following conclusions is best supported by the information provided?
O Because there was a change in energy, the equation must not be balanced
O the overall mass of the system increased when the reaction occurred
O This reaction required the addition of energy from the surroundings
O During the reaction the energy was converted to mass, and, therefore, was lost

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2

You know the right answer?
The diagram provided here shows the reactants and the products of a common chemical reaction. The li...
Questions


Mathematics, 27.10.2020 04:20


Arts, 27.10.2020 04:20


Chemistry, 27.10.2020 04:20

Physics, 27.10.2020 04:20


Social Studies, 27.10.2020 04:20



Biology, 27.10.2020 04:20



Physics, 27.10.2020 04:20

World Languages, 27.10.2020 04:20

Mathematics, 27.10.2020 04:20





