
Chemistry, 29.05.2021 21:00 moose12002
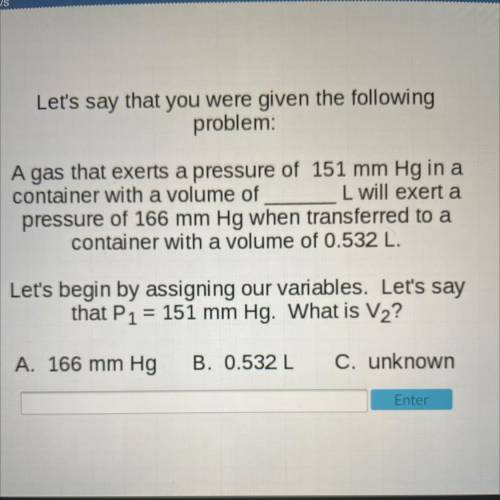
Let's say that you were given the following
problem:
A gas that exerts a pressure of 151 mm Hg in a
container with a volume of L will exert a
pressure of 166 mm Hg when transferred to a
container with a volume of 0.532 L.
Let's begin by assigning our variables. Let's say
that P1 = 151 mm Hg. What is V2?

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1
You know the right answer?
Let's say that you were given the following
problem:
A gas that exerts a pressure of 151 mm H...
A gas that exerts a pressure of 151 mm H...
Questions




Health, 03.07.2019 17:00

Mathematics, 03.07.2019 17:00


Mathematics, 03.07.2019 17:00



Social Studies, 03.07.2019 17:00



Mathematics, 03.07.2019 17:00

Health, 03.07.2019 17:00






History, 03.07.2019 17:00



