
An engine operates on a Carnot cycle that uses 1mole of an ideal gas as the
working substance and operates from a most compressed stage of 100 Nm and
327 K. It expands isothermally to a pressure of 90 Nm and then adiabatically to a
most expanded stage of 27 K. Calculate the AU, 9, and w for each step. Calculate
the net work done and the efficiency of the cycle [Cv, m for the gas) is 25 J/k/mol.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
In which layer of earth do most earthauakes occur a_ inner core b_outer core c_mantle d_crust
Answers: 1

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1

Chemistry, 23.06.2019 00:30
Which of the following best describes technology a. something created for only scientists to use b.the method of thinking that scientists use. c.the application of engineering to create useful products. c. a scientific idea
Answers: 1

Chemistry, 23.06.2019 01:00
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2
You know the right answer?
An engine operates on a Carnot cycle that uses 1mole of an ideal gas as the
working substance and o...
Questions


Mathematics, 05.11.2020 03:00


English, 05.11.2020 03:00

Mathematics, 05.11.2020 03:00


Business, 05.11.2020 03:00



Chemistry, 05.11.2020 03:00

English, 05.11.2020 03:00

Mathematics, 05.11.2020 03:00

Chemistry, 05.11.2020 03:00


English, 05.11.2020 03:00

Mathematics, 05.11.2020 03:00


Mathematics, 05.11.2020 03:00


Social Studies, 05.11.2020 03:00

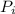 = 100 N·m
= 100 N·m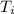 = 327 K
= 327 K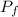 = 90 N·m
= 90 N·m

