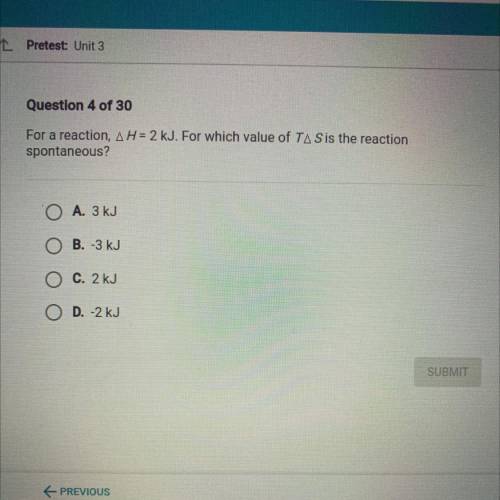
For a reaction, AH = 2 kJ. For which value of TA Sis the reaction
spontaneous?
...

Chemistry, 31.05.2021 22:00 moningersavannah
For a reaction, AH = 2 kJ. For which value of TA Sis the reaction
spontaneous?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 20:00
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1
You know the right answer?
Questions

Computers and Technology, 04.04.2020 13:01



Business, 04.04.2020 13:01


Chemistry, 04.04.2020 13:01

Mathematics, 04.04.2020 13:01

Geography, 04.04.2020 13:01











German, 04.04.2020 13:01



