
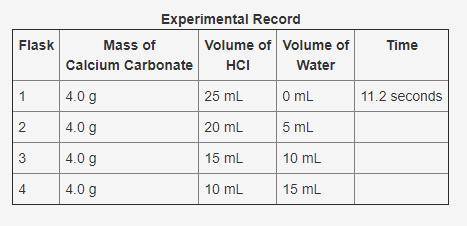
In an experiment, calcium carbonate reacted with different volumes of hydrochloric acid in water. One of the products formed during the experiment was carbon dioxide. The time taken for 0.89 mL of carbon dioxide to form was recorded. A partial record of the experiment is shown. Based on your knowledge of factors that affect the rates of chemical reactions, predict the trend in the last column of the experimental record. Use complete sentences to explain the trend you predicted. You do not have to determine exact values for time; just describe the trend you would expect (increase or decrease) and why it occurs.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
The big bang nucleosynthesis theory states that elements were produced in the first few minutes of the big bang while elements have their origins in the interiors of stars, forming much later in the history of the universe.
Answers: 1

Chemistry, 22.06.2019 08:30
How does the principle of electromagnetism explain the interaction between earth’s magnetic field and the solar wind?
Answers: 1

Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2

You know the right answer?
In an experiment, calcium carbonate reacted with different volumes of hydrochloric acid in water. On...
Questions

Computers and Technology, 15.10.2020 08:01


Social Studies, 15.10.2020 08:01






Biology, 15.10.2020 08:01

History, 15.10.2020 08:01

History, 15.10.2020 08:01



English, 15.10.2020 08:01

Computers and Technology, 15.10.2020 08:01


Computers and Technology, 15.10.2020 08:01





