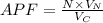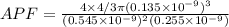Consider a hypothetical metal that has a density of 3.55 g/cm3, an atomic weight of 40.48 g/mol, and an atomic radius of 0.135 nm. Compute the atomic packing factor if the unit cell has tetragonal symmetry, values for the a and c lattice parameters are 0.545 and 0.255, respectively.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:40
Asingle atom of an element has 21 neutrons, 20 electrons, and 20 protons. which element is it? ok o z
Answers: 1

Chemistry, 22.06.2019 00:30
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 06:20
If i can still dissolve more sugar into the solution at a certain temperature what would i call that solution
Answers: 3
You know the right answer?
Consider a hypothetical metal that has a density of 3.55 g/cm3, an atomic weight of 40.48 g/mol, and...
Questions





Biology, 17.07.2019 07:00


Biology, 17.07.2019 07:00

Health, 17.07.2019 07:00

Biology, 17.07.2019 07:00


Mathematics, 17.07.2019 07:00

History, 17.07.2019 07:00

Business, 17.07.2019 07:00

History, 17.07.2019 07:00


Chemistry, 17.07.2019 07:00


Social Studies, 17.07.2019 07:00