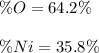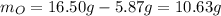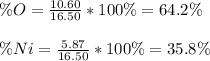Chemistry, 03.06.2021 23:30 kaitlynmoore42
When a 16.50 g sample containing nickel and oxygen is analyzed, 5.87 g of nickel are found. What is the percent composition of this mineral?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 23:00
Which type of intermolecular attractions holds ammonia molecules together with other ammonia molecules?
Answers: 3

Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
You know the right answer?
When a 16.50 g sample containing nickel and oxygen is analyzed, 5.87 g of nickel are found. What is...
Questions

English, 31.05.2021 18:20

Social Studies, 31.05.2021 18:20







Mathematics, 31.05.2021 18:20

Computers and Technology, 31.05.2021 18:20

Mathematics, 31.05.2021 18:20

Mathematics, 31.05.2021 18:20




Physics, 31.05.2021 18:20

English, 31.05.2021 18:20

English, 31.05.2021 18:20