
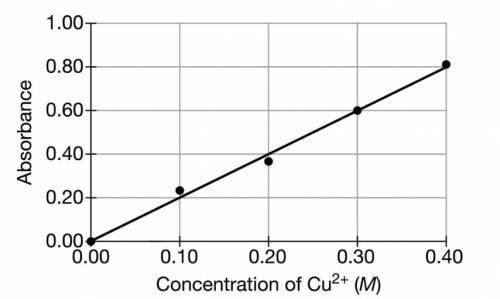
A student is given a sample of CuSO4(s) that contains a solid impurity that is soluble and colorless. The student wants to determine the amount of CuSO4 in the sample and decides to use a spectrophotometer. First, the student prepares a calibration graph by measuring the absorbances of CuSO4(aq) solutions of known concentrations. The graph is shown below.
(a) The student dissolves the entire impure sample of CuSO4(s) in enough distilled water to make 100.mL of solution. Then the student measures the absorbance of the solution and observes that it is 0.30. Determine the concentration of CuSO4(aq) in the solution.
(b) Calculate the number of moles of CuSO4 that were in the impure sample of CuSO4(s).
(c) In addition to the number of moles of CuSO4 calculated in part (b), what other quantity must be measured in order to calculate the mass percentage of CuSO4 in the impure sample of CuSO4(s)?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2


Chemistry, 22.06.2019 20:00
If one fission reaction of a uranium-235 atom produced two neutrons, how many neutrons would be released if the chain reaction occurred three more times?
Answers: 1
You know the right answer?
A student is given a sample of CuSO4(s) that contains a solid impurity that is soluble and colorless...
Questions

History, 20.10.2020 08:01

English, 20.10.2020 08:01

Chemistry, 20.10.2020 08:01

Mathematics, 20.10.2020 08:01

History, 20.10.2020 08:01


History, 20.10.2020 08:01


Social Studies, 20.10.2020 08:01

English, 20.10.2020 08:01


Mathematics, 20.10.2020 08:01


Mathematics, 20.10.2020 08:01

Mathematics, 20.10.2020 08:01







