Question 21
Which equation is a balanced chemical equation?
A
2NaOH + CaBr2 + Ca(OH)2 +...

Chemistry, 05.06.2021 01:00 deontehiggins42
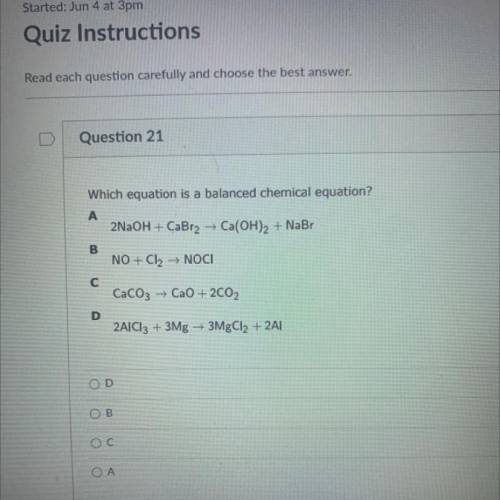
Question 21
Which equation is a balanced chemical equation?
A
2NaOH + CaBr2 + Ca(OH)2 + NaBr
B
NO + Cl2 → NOCI
с
CaCO3 + CaO + 2CO2
D
2AlCl3 + 3Mg + 3MgCl2 + 2A|
D
B
Ос
ОА

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3
You know the right answer?
Questions






English, 16.12.2021 21:10


Mathematics, 16.12.2021 21:10


Mathematics, 16.12.2021 21:10


English, 16.12.2021 21:10


Mathematics, 16.12.2021 21:10

Mathematics, 16.12.2021 21:10


Mathematics, 16.12.2021 21:10





