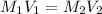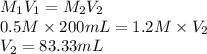Chemistry, 05.06.2021 03:50 brownboimexican
Sample of 200mls of 0.5 sulphuric acid, was asked to produce 1.2M of the new solution. Calculate the volume of the new solution

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2
You know the right answer?
Sample of 200mls of 0.5 sulphuric acid, was asked to produce 1.2M of the new solution. Calculate the...
Questions


















Social Studies, 06.12.2019 01:31


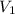 = 200 mL,
= 200 mL, 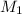 = 0.5 M
= 0.5 M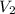 = ?,
= ?, 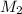 = 1.2 M
= 1.2 M