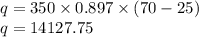Chemistry, 06.06.2021 19:00 hhgjhbmbhg266
Calculate the amount of heat associated with cooling a 350.0 g aluminum bar from 70.0 oC to 25.0 oC. The specific heat of aluminum is 0.897 J/g oC. (–14,127.75 J)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 02:00
What is the maximum number of electrons that an atomic orbital can contain?
Answers: 1

Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2
You know the right answer?
Calculate the amount of heat associated with cooling a 350.0 g aluminum bar from 70.0 oC to 25.0 oC....
Questions








Mathematics, 09.12.2021 04:50


Mathematics, 09.12.2021 04:50



Business, 09.12.2021 04:50


Mathematics, 09.12.2021 04:50

Mathematics, 09.12.2021 04:50

English, 09.12.2021 04:50


Mathematics, 09.12.2021 04:50