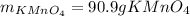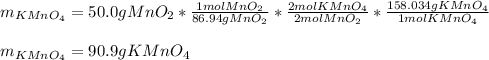Chemistry, 07.06.2021 05:50 lordcaos066
A company manufacturing KMnO4 wants to obtain the highest yield possible. Two of their research scientists are working on a technique to increase the yield.
Both scientists started with 50.0 g of manganese oxide (MnO2).
What is the theoretical yield of potassium permanganate when starting with this 50.0 g MnO2?
The equation for the production of potassium permanganate is as follows:
2 MnO2 + 2 KOH + O2 → 2 KMnO4 + H2
You must show all work to receive full credit.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Smog is the term used to describe the combination of fog and smoke
Answers: 1

Chemistry, 22.06.2019 01:40
C3h8o3 - glycerol major species present when dissolved in water
Answers: 2

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1
You know the right answer?
A company manufacturing KMnO4 wants to obtain the highest yield possible. Two of their research scie...
Questions











Mathematics, 26.07.2019 21:10



Mathematics, 26.07.2019 21:10