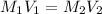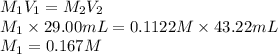Chemistry, 07.06.2021 15:30 biggiecheese93
A 29.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1122 M KOH solution. A volume of 43.22 mL of KOH was required to reach the equivalence point. What is the concentration of the unknown H2SO4 solution? Express your answer using four significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution? a. 3.88 m, b. 1.03 m, c. 1.5 m, d. 15.5 m
Answers: 3

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1

Chemistry, 22.06.2019 19:20
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3
You know the right answer?
A 29.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1122 M KOH solu...
Questions

History, 27.01.2021 23:10


English, 27.01.2021 23:10



Mathematics, 27.01.2021 23:10

Mathematics, 27.01.2021 23:10

Mathematics, 27.01.2021 23:10

Mathematics, 27.01.2021 23:10

Mathematics, 27.01.2021 23:10

Geography, 27.01.2021 23:10

Biology, 27.01.2021 23:10




Chemistry, 27.01.2021 23:10




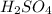 solution is 0.167 M.
solution is 0.167 M.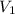 = 29.00 mL,
= 29.00 mL, 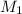 = ?
= ?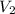 = 43.22 mL,
= 43.22 mL, 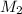 = 0.1122 M
= 0.1122 M