
For the equilibrium that exists in an aqueous solution of nitrous acid (HNO2, a weak acid), the equilibrium constant expression is:
a. K = [ H+] [NO2-] / [HNO2]
b. K = [ H+] [N] [O]2 / [HNO2]
c. K = [ H+] [NO2-] / [HNO2]
d. K = [H+]2 [NO2-] / [HNO2]
e. None of these

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 23.06.2019 01:30
Will a solution form when the solvent and solute are both nonpolar? a. not likely b. never c. most likely
Answers: 1

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1
You know the right answer?
For the equilibrium that exists in an aqueous solution of nitrous acid (HNO2, a weak acid), the equi...
Questions



Mathematics, 27.09.2019 00:50

Mathematics, 27.09.2019 00:50

Biology, 27.09.2019 00:50

History, 27.09.2019 00:50



Physics, 27.09.2019 00:50




Mathematics, 27.09.2019 00:50

Mathematics, 27.09.2019 00:50

Geography, 27.09.2019 00:50


Mathematics, 27.09.2019 00:50

Mathematics, 27.09.2019 00:50


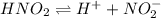
![K = \frac{[H^{+}][NO^{-}_{2}]}{[HNO_{2}]}](/tpl/images/1365/7220/b80db.png)


