
Chemistry, 08.06.2021 08:00 emilaw3233
The reaction of bromine gas with chlorine gas, shown here, has a Keq value of 7.20 at 200°C. If a closed vessel was charged with the two reactants, each at an initial concentration of 0.200 M, but with no initial concentration of BrCl, what would be the equilibrium concentration of Br2, Cl2 and BrCl(g)? Br2(g) + Cl2(g) ↔ 2BrCl(g) K = 7.20

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
The reaction of bromine gas with chlorine gas, shown here, has a Keq value of 7.20 at 200°C. If a cl...
Questions


Mathematics, 16.07.2019 00:00



Mathematics, 16.07.2019 00:00

Arts, 16.07.2019 00:00

History, 16.07.2019 00:00



Social Studies, 16.07.2019 00:00

Mathematics, 16.07.2019 00:00

History, 16.07.2019 00:00

Health, 16.07.2019 00:00


Mathematics, 16.07.2019 00:00

History, 16.07.2019 00:00


Mathematics, 16.07.2019 00:00

Mathematics, 16.07.2019 00:00

![[Cl_2]=[Br_2]=0.856M](/tpl/images/1366/5138/5290e.png)
![[BrCl]=0.229M](/tpl/images/1366/5138/704d0.png)
![Keq=\frac{[BrCl]^2}{[Cl_2][Br_2]}](/tpl/images/1366/5138/7a521.png)
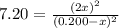
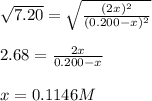
![[Cl_2]=[Br_2]=0.200M-0.1146M=0.856M](/tpl/images/1366/5138/7f591.png)
![[BrCl]=2*0.1146M=0.229M](/tpl/images/1366/5138/64b19.png)



