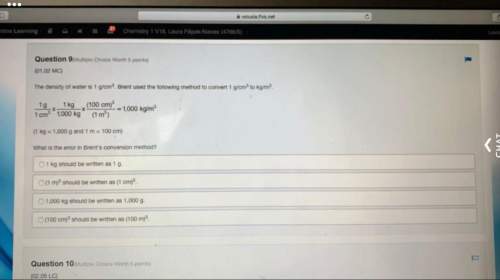
Chemistry, 08.06.2021 23:30 nellydeb7736
Hot lead with a mass of 200.0 g of (Specific heat
of Pb = 0.129 Jig'C) at 176.4°C was dropped into
a calorimeter containing an unknown volume of
water. The temperature of the water increased
from 21.7°C to 56.4°C. What volume of water is
in the calorimeter?
a. 11. 32 mL
b. 21.32 mL
C. 12.23 mL
d. 31.21 mL

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2


Chemistry, 23.06.2019 09:00
Water is a highly important natural resource. which of these would be the best method to conserve water? a) drinking bottled water b) monitoring the ph of rivers c) treating and re-using wastewater d) testing nitrate levels in groundwater
Answers: 1
You know the right answer?
Hot lead with a mass of 200.0 g of (Specific heat
of Pb = 0.129 Jig'C) at 176.4°C was dropped into<...
Questions



Mathematics, 01.11.2020 05:20


Business, 01.11.2020 05:30

Social Studies, 01.11.2020 05:30

History, 01.11.2020 05:30

English, 01.11.2020 05:30


Social Studies, 01.11.2020 05:30

Mathematics, 01.11.2020 05:30


English, 01.11.2020 05:30

Spanish, 01.11.2020 05:30


Health, 01.11.2020 05:30

Mathematics, 01.11.2020 05:30


English, 01.11.2020 05:30