Asnwer?
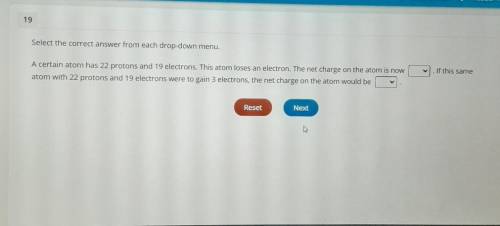
A certain atom has 22 protons and 19 electrons. this atom loses an electron.
the net o...

Chemistry, 10.06.2021 01:50 haileyhale5
Asnwer?
A certain atom has 22 protons and 19 electrons. this atom loses an electron.
the net of the aton is now ___.(4+,1+,0,1-,4-)
if this same atom with 22 protons and 19 electrons were to gain 3 electrons, the net charge on the atom would be___.(3+,2+,0,2-,3-)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 23.06.2019 05:40
The independent variable in an experiment will be the variable that you o a) change ob) hold constant ng c) observe for changes
Answers: 2

Chemistry, 23.06.2019 09:00
Avogradoa number was calculated by determining the number of atoms in?
Answers: 1
You know the right answer?
Questions

Biology, 29.10.2020 20:20








Arts, 29.10.2020 20:20

Mathematics, 29.10.2020 20:20


Mathematics, 29.10.2020 20:20

Mathematics, 29.10.2020 20:20



Mathematics, 29.10.2020 20:20

Mathematics, 29.10.2020 20:20



Mathematics, 29.10.2020 20:20



