
Chemistry, 10.06.2021 04:00 djtblaster
Given two equations representing reactions: which type of reaction is represented by each of these equations?
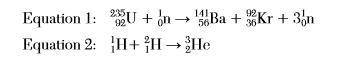

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:20
Calculate the molarity of the solution. 6.02 x 1022 molecules of hci (molecular weight = 36.5 g/mole) in 2.0 liters of water m
Answers: 1

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1
You know the right answer?
Given two equations representing reactions:
which type of reaction is represented by each of these...
Questions

Mathematics, 20.10.2020 22:01

Biology, 20.10.2020 22:01


Computers and Technology, 20.10.2020 22:01



Social Studies, 20.10.2020 22:01


English, 20.10.2020 22:01

English, 20.10.2020 22:01





Mathematics, 20.10.2020 22:01







