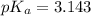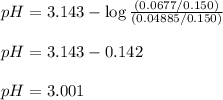Chemistry, 11.06.2021 22:30 joshtassler15
A buffer solution contains 0.475 M nitrous acid and 0.302 M sodium nitrite . If 0.0224 moles of potassium hydroxide are added to 150 mL of this buffer, what is the pH of the resulting solution

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1

Chemistry, 22.06.2019 22:00
All of the following are homogeneous mixtures except a) sugar dissolved in water. b) orange juice. c) coffee with cream. d) household vinegar. e) apple juice
Answers: 1

Chemistry, 23.06.2019 03:30
If 2 molecules of one reactant combine with 3 molecules of another to produce 5 molecules of a product, then what is the representation of the reaction?
Answers: 1
You know the right answer?
A buffer solution contains 0.475 M nitrous acid and 0.302 M sodium nitrite . If 0.0224 moles of pota...
Questions


Mathematics, 28.09.2019 00:00


History, 28.09.2019 00:00




Mathematics, 28.09.2019 00:00


Mathematics, 28.09.2019 00:00

Mathematics, 28.09.2019 00:00

Mathematics, 28.09.2019 00:00




Mathematics, 28.09.2019 00:00



Mathematics, 28.09.2019 00:00

Social Studies, 28.09.2019 00:00

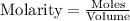 ......(1)
......(1)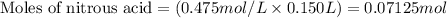
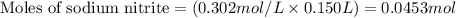
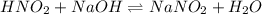
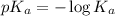 ......(2)
......(2)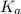 for nitrous acid =
for nitrous acid = 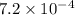
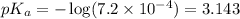
![pH=pK_a+ \log \frac{\text{[conjugate base]}}{\text{[acid]}}](/tpl/images/1372/0136/096b8.png) .......(3)
.......(3)![[NaNO_2]=\frac{0.0677}{0.150}](/tpl/images/1372/0136/ed413.png)
![[HNO_2]=\frac{0.04885}{0.150}](/tpl/images/1372/0136/86d5f.png)