Chemistry, 11.06.2021 22:50 tyneshiajones124
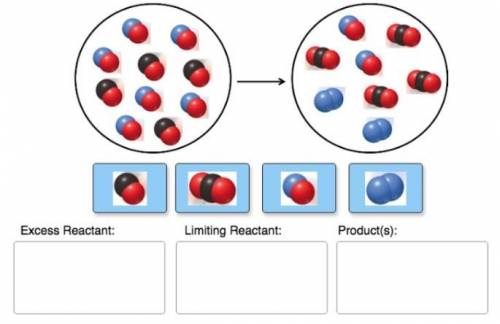
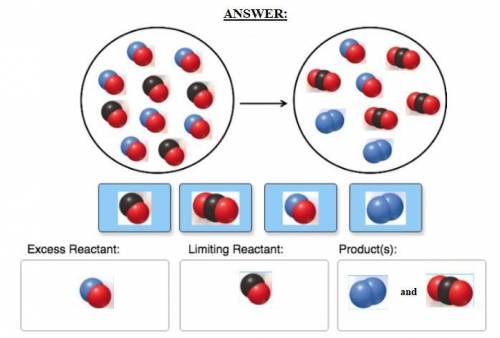
Consider the reaction of NO and CO to form N2 and CO2, according to the balanced equation: 2 NO (g) + 2 CO (g) → N2 (g) + 2 CO2 (g) Identify the excess reactant, the limiting reactant, and the product(s) using the molecular art. (Black spheres are carbon, blue spheres are nitrogen, and red spheres are oxygen.)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Which orbitals form a pi bond? a.the s orbital and three p orbitals b.the s orbital and two p orbitals c.overlapping p orbitals d.overlapping hybrid orbitals
Answers: 2

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 16:00
Uranium can supply energy for the worlds electricity without admitting harmful greenhouse gases which of these statements best describes an outcome of uranium mining
Answers: 1

Chemistry, 22.06.2019 19:00
How many moles of cu are needed to react with 5.8 moles of agno3? cu + 2 agno3 → cu(no3)2 + 2 ag
Answers: 3
You know the right answer?
Consider the reaction of NO and CO to form N2 and CO2, according to the balanced equation: 2 NO (g)...
Questions





Computers and Technology, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00


Chemistry, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00

Physics, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00


Physics, 08.10.2021 14:00

History, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00

Law, 08.10.2021 14:00


English, 08.10.2021 14:00

History, 08.10.2021 14:00

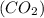 compound
compound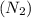
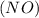 compound
compound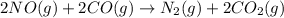
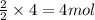 of NO
of NO