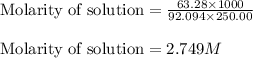Chemistry, 15.06.2021 14:00 msladycie8831
Glycerol. C3HgO3, is a substance used extensively in the manufacture of cosmetics, foodstuffs, antifreeze, and plastics. Glycerol is a water-soluble liquid
with a density of 1.2656 g/mL at 15 °C. Calculate the molarity of a solution of glycerol made by dissolving 50.000 mL glycerol at 15 °C in enough water to
make 250.00 mL of solution. The molecular weight of C3HgO3 is 92.094 amu.
O A 0.6871
O B. 3.600
O C. 63.28
O 0.92.10
O E. 2.749

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 10:00
Why is the structure of molecule important to its function?
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 23.06.2019 01:10
Volume is a measurement of how fast particles of a substance are moving
Answers: 3
You know the right answer?
Glycerol. C3HgO3, is a substance used extensively in the manufacture of cosmetics, foodstuffs, antif...
Questions



Mathematics, 05.03.2020 07:06


Mathematics, 05.03.2020 07:06

English, 05.03.2020 07:07


Computers and Technology, 05.03.2020 07:07



Biology, 05.03.2020 07:07


Biology, 05.03.2020 07:07


Mathematics, 05.03.2020 07:07




Mathematics, 05.03.2020 07:08

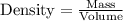 ......(1)
......(1)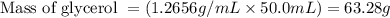
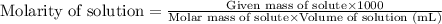 .....(2)
.....(2)