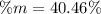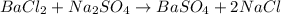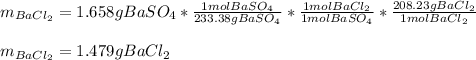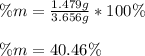Chemistry, 16.06.2021 01:00 chodister353
A mixture of BaCl2 and NaCl is analyzed by precipitating all the barium as BaSO4. After addition of an excess of Na2SO4 to a 3.656-g sample of the mixture, the mass of precipitate collected is 1.658 g. What is the mass percentage of barium chloride in the mixture

Answers: 3


Another question on Chemistry

Chemistry, 23.06.2019 01:30
What happens to the concentration of hydronium ions as the ph of a solution increases? a. hydronium ion concentration stays the same b. hydronium ion concentration decreases c. hydronium ion concentration increases
Answers: 1

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3

Chemistry, 23.06.2019 08:40
The half-life of a certain element is 100 days. how many half-lives will it be before only one eighth of this elementremains?
Answers: 1

Chemistry, 23.06.2019 09:20
Due tomorrow which would have a lower ph, a 0.1 m solution of a strong base or a weak base? why? which would have a higher ph, a 0.1 m solution of a strong base or a weak base? why?
Answers: 3
You know the right answer?
A mixture of BaCl2 and NaCl is analyzed by precipitating all the barium as BaSO4. After addition of...
Questions



















Mathematics, 14.07.2020 23:01