
Chemistry, 11.10.2019 19:30 rowdycar313p0ao5k
What is the theoretical yield of ammonia that can be obtained from the reaction of 10.0 g of h2 and excess n2?
n2 + 3h2 → 2nh3
a. 90.0 g
b. 97.1 g
c. 56.3 g
d. 48.6 g
e. 28.4 g

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Select the correct text in the passage. which sentences describe examples of sustainable living? i live in an old apartment building downtown, but my company is based in an office park on the outskirts of the city. i drive an old car that needs to be replaced. i plan to buy a hybrid for better gas mileage, but for now i am able to carpool with a couple of friends from work. the drive to the office park is about 45 minutes each way, but we do get to work in a modern building. the architects just received a leed certification for the design.
Answers: 3


Chemistry, 23.06.2019 06:00
Amanda pushes a box across the room with a force of 30 n. it accelerates at 5 m/s/s. what is the mass of the box? * 6 kg 1.16 kg 30 kg 5kg
Answers: 2

You know the right answer?
What is the theoretical yield of ammonia that can be obtained from the reaction of 10.0 g of h2 and...
Questions



Computers and Technology, 15.04.2020 07:20

History, 15.04.2020 07:20

Mathematics, 15.04.2020 07:20

Mathematics, 15.04.2020 07:20

Mathematics, 15.04.2020 07:20





Mathematics, 15.04.2020 07:20

Mathematics, 15.04.2020 07:21


Biology, 15.04.2020 07:21



Social Studies, 15.04.2020 07:21


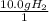 ×
×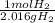 ×
×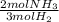 ×
×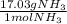 =56.3gNH↓3
=56.3gNH↓3

