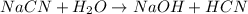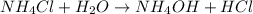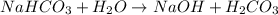Chemistry, 18.06.2021 02:00 alananicoleee
State whether 0.1 M solutions of each of the following salts are acidic, basic, or neutral. Explain your reasoning for each by writing a balanced net ionic equation to describe the behavior of each nonneutral salt in water: NaCN, KNO3, NH4Cl, NaHCO3, and Na3PO4.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What is the mass of each element in a 324.8 sample of co2
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1

Chemistry, 22.06.2019 22:30
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1
You know the right answer?
State whether 0.1 M solutions of each of the following salts are acidic, basic, or neutral. Explain...
Questions


Biology, 12.07.2019 03:00

Biology, 12.07.2019 03:00




History, 12.07.2019 03:00



Business, 12.07.2019 03:00


Biology, 12.07.2019 03:00




Computers and Technology, 12.07.2019 03:00

Mathematics, 12.07.2019 03:00


Mathematics, 12.07.2019 03:00

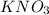 is a neutral salt
is a neutral salt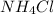 is an acidic salt
is an acidic salt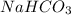 is a basic salt
is a basic salt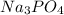 is a neutral salt
is a neutral salt