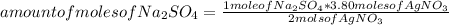Silver sulfate is precipitated from solution according to the following reaction 2 AgNO3 (aq) Na2SO4 (aq) --> Ag2SO4 (s) 2 NaNO3 (aq) If 3.80 moles of AgNO3 and 3.05 moles of Na2SO4 are present initially, how many moles of excess reactant will remain after precipitation is complete

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:00
The diagram shows the positions of the sun, moon and earth during spring tides, when the high tides are at their highest and low tides at their lowest. what is it about these positions that causes these high and low tides?
Answers: 3

Chemistry, 22.06.2019 08:00
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 19:00
Avolleyball player hit a ball with a mass of 0.25 kg. the average acceleration of the ball is 15.5 m/s². how much force did the volleyball player apply to the ball? 62.0 n 3.87 n 62.0 m/s² 3.87 m/s²
Answers: 2
You know the right answer?
Silver sulfate is precipitated from solution according to the following reaction 2 AgNO3 (aq) Na2SO4...
Questions


Social Studies, 08.01.2021 19:00

Chemistry, 08.01.2021 19:00

Mathematics, 08.01.2021 19:00


Mathematics, 08.01.2021 19:00




Mathematics, 08.01.2021 19:00

Computers and Technology, 08.01.2021 19:00

Mathematics, 08.01.2021 19:00


Health, 08.01.2021 19:00


Mathematics, 08.01.2021 19:00

Mathematics, 08.01.2021 19:00


Physics, 08.01.2021 19:00

Arts, 08.01.2021 19:00