Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Consider the balanced equation below. n2h4 + 2h2o2 n2 + 4h2o what are the mole ratios of hydrazine (n2h4) to hydrogen peroxide (h2o2) and hydrazine to water? 1: 2 and 1: 4 1: 3 and 1: 4 1: 2 and 3: 5 1: 3 and 3: 5
Answers: 3

Chemistry, 21.06.2019 15:30
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 21.06.2019 18:00
Now consider the reaction when 45.0 g naoh have been added. what amount of naoh is this, and what amount of fecl3 can be consumed by it?
Answers: 3

Chemistry, 22.06.2019 12:00
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1
You know the right answer?
How much water can be produced when 18 grams of pentane is burned in air? C5H12 + 8 02 --> 5 CO2...
Questions

Biology, 24.03.2021 18:30


Mathematics, 24.03.2021 18:30

Mathematics, 24.03.2021 18:30



Mathematics, 24.03.2021 18:30



Health, 24.03.2021 18:30



Mathematics, 24.03.2021 18:30

Biology, 24.03.2021 18:30

Mathematics, 24.03.2021 18:30




English, 24.03.2021 18:30

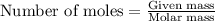 ......(1)
......(1)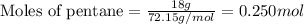
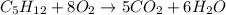
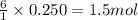 of water
of water