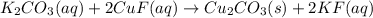Aqueous potassium carbonate was mixed with aqueous copper (1) fluoride and a crystallized copper (1) carbonate product was formed. A crystalized product is a solid. The other product, potassium fluoride, remains dissolved in solution. Consider the other product and it’s phase, and then write the balanced molecular equation for this precipitation reaction. Express your answer as a chemical equation including phases. Type an underscore (_) or a carat (^) to add subscripts and superscripted more quickly.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

Chemistry, 22.06.2019 14:50
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1


Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3
You know the right answer?
Aqueous potassium carbonate was mixed with aqueous copper (1) fluoride and a crystallized copper (1)...
Questions


History, 15.04.2021 04:00


Mathematics, 15.04.2021 04:00

Mathematics, 15.04.2021 04:00

Mathematics, 15.04.2021 04:00

Biology, 15.04.2021 04:00

Mathematics, 15.04.2021 04:00

Mathematics, 15.04.2021 04:00

Mathematics, 15.04.2021 04:00


Social Studies, 15.04.2021 04:00

Mathematics, 15.04.2021 04:00

Mathematics, 15.04.2021 04:00



Mathematics, 15.04.2021 04:00

Chemistry, 15.04.2021 04:00


Chemistry, 15.04.2021 04:00