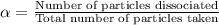Zinc sulfate is a 2-ion electrolyte,
dissociating 40% in a
certain concentration. Calculate i...

Chemistry, 21.06.2021 14:00 atsuedem974
Zinc sulfate is a 2-ion electrolyte,
dissociating 40% in a
certain concentration. Calculate its
dissociation (i) factor.
On the basis of 40% dissociation, 100
particles of zinc sulfate
will yield:
40zinc ions
40 sulfate ions
60undissociated particles
Jo 11:03

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 08:50
Why are enzymes important to cells? they bring about chemical reactions. they provide structural support. they form the two layers of membranes. they store large quantities of energy.
Answers: 2

Chemistry, 23.06.2019 10:00
What is the density, d, of a substance with a volume of v = 12.5 cm3 and a mass of m = 74.4 g ?
Answers: 1

Chemistry, 23.06.2019 10:30
Ethyl alcohol, also known as ethanol, has a density of 0.79 g/ml. what is the volume, in quarts, of 1.95 kg of this alcohol?
Answers: 2

Chemistry, 23.06.2019 14:00
Beaker a contains 100 ml of 1.5 mammonia. beaker b contains 100 ml of 0.50m ammonia. how does the solution in beaker a compare to the solution in beaker b?
Answers: 2
You know the right answer?
Questions


Physics, 31.08.2020 03:01



Mathematics, 31.08.2020 03:01

Computers and Technology, 31.08.2020 03:01

Computers and Technology, 31.08.2020 03:01




English, 31.08.2020 03:01

English, 31.08.2020 03:01




Chemistry, 31.08.2020 03:01



Advanced Placement (AP), 31.08.2020 03:01

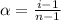
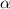 = degree of dissociation = 40% = 0.40
= degree of dissociation = 40% = 0.40