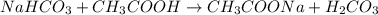Chemistry, 21.06.2021 17:50 danielcano12281621
PLEASE HELP
The average number of drops of NaHCO3 in the titration lab was 142. Calculate the number of moles of NaCHO3 that were required to neutralize the CH3COOH in the vinegar.
- 1 mole of acetic acid was required for 2 moles of sodium bicarbonate
- 1 mole of acetic acid was required for 1 mole of sodium bicarbonate
- 2 mole of acetic acid was required for 1 mole of sodium bicarbonate
- 1 mole of acetic acid was required for 3 mole of sodium bicarbonate

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 23.06.2019 01:30
Concentrations expressed as a percent by mass are useful when the solute is a a. liquid b. gas c. solid
Answers: 1

Chemistry, 23.06.2019 09:30
The mass of a proton is approximately equal to the mass of
Answers: 1
You know the right answer?
PLEASE HELP
The average number of drops of NaHCO3 in the titration lab was 142. Calculate the numbe...
Questions


English, 26.10.2021 23:40


Mathematics, 26.10.2021 23:40

English, 26.10.2021 23:40

Mathematics, 26.10.2021 23:40



Geography, 26.10.2021 23:40




Mathematics, 26.10.2021 23:40


English, 26.10.2021 23:40



Social Studies, 26.10.2021 23:40