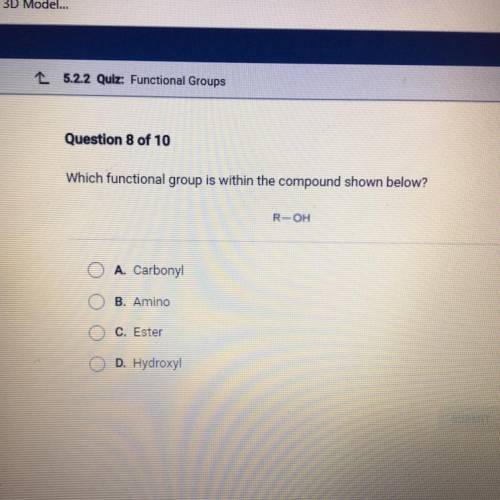
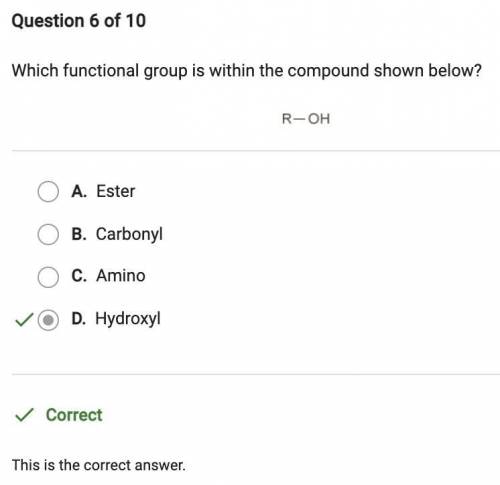
Question 8 of 10
Which functional group is within the compound shown below?
R-OH
A. Car...


Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
You know the right answer?
Questions

History, 17.06.2020 02:57


Mathematics, 17.06.2020 02:57

English, 17.06.2020 02:57

Mathematics, 17.06.2020 02:57

Mathematics, 17.06.2020 02:57


Mathematics, 17.06.2020 02:57

Mathematics, 17.06.2020 02:57

English, 17.06.2020 02:57

History, 17.06.2020 02:57


Mathematics, 17.06.2020 02:57

Mathematics, 17.06.2020 02:57

Mathematics, 17.06.2020 02:57

Mathematics, 17.06.2020 02:57

English, 17.06.2020 02:57


Mathematics, 17.06.2020 02:57

Mathematics, 17.06.2020 02:57